Examples of chemical changes4/6/2023 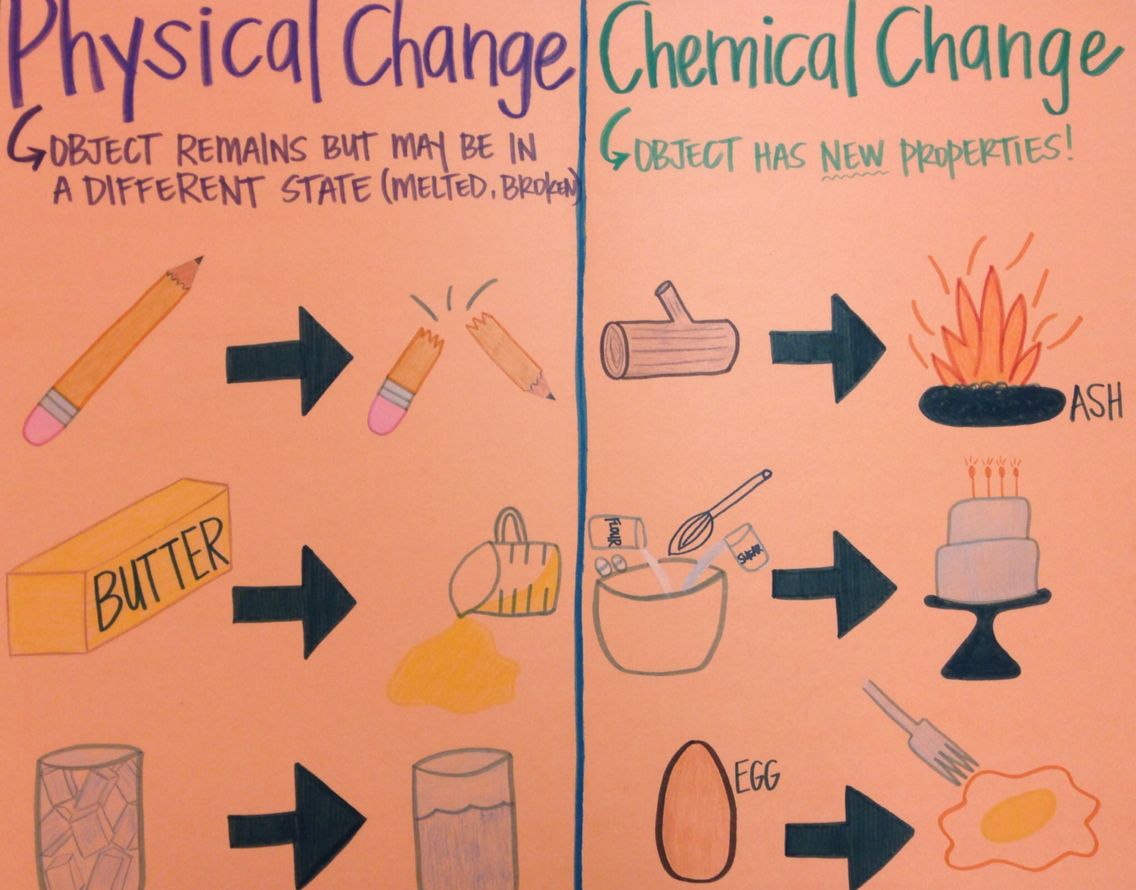
In his book Traité élémentaire de chimie (1789 Elementary Treatise on Chemistry), Lavoisier identified 33 “elements”-substances not broken down into simpler entities. The influence of French chemist Antoine-Laurent Lavoisier was especially notable, in that his insights confirmed the importance of quantitative measurements of chemical processes. 
The identification of oxygen in the 18th century by Swedish chemist Carl Wilhelm Scheele and English clergyman Joseph Priestley had particular significance. The first substantive studies in this area were on gases. Get a Britannica Premium subscription and gain access to exclusive content. Development of the concept of a chemical reaction had a primary role in defining the science of chemistry as it is known today. It had its origins in early experiments that classified substances as elements and compounds and in theories that explained these processes. The concept of a chemical reaction dates back about 250 years. By this, we know that a chemical change or reaction has occurred. However, if water, as ice, liquid, or vapour, encounters sodium metal (Na), the atoms will be redistributed to give the new substances molecular hydrogen (H 2) and sodium hydroxide (NaOH). No matter what its physical state, water (H 2O) is the same compound, with each molecule composed of two atoms of hydrogen and one atom of oxygen. 
If a physical change occurs, the physical properties of a substance will change, but its chemical identity will remain the same. Physical changes include changes of state, such as ice melting to water and water evaporating to vapour. Melting ice Wayne Dionne/© New Hampshire Division of Travel and Tourism DevelopmentĬhemical reactions must be distinguished from physical changes. Chain reactions and photolysis reactions are examples of classification by reaction mechanism, which provides details on how atoms are shuffled and reassembled in the formation of products. Examples of classification by reaction outcome include decomposition, polymerization, substitution, and elimination and addition reactions. Classification by types of reactants include acid-base reactions and oxidation-reduction reactions, which involve the transfer of one or more electrons from a reducing agent to an oxidizing agent. Cake batter rising is caused by a gas-forming reaction between an acid and baking soda (sodium hydrogen carbonate). Many reactions produce a gas such as carbon dioxide, hydrogen sulfide, ammonia, or sulfur dioxide. /TC_608336-examples-of-physical-changes-5aa986371f4e1300371ebebb.png)
Often a given reaction can be placed in two or even three categories, including gas-forming and precipitation reactions. SpaceNext50 Britannica presents SpaceNext50, From the race to the Moon to space stewardship, we explore a wide range of subjects that feed our curiosity about space!Ĭhemists classify chemical reactions in a number of ways: by type of product, by types of reactants, by reaction outcome, and by reaction mechanism.Learn about the major environmental problems facing our planet and what can be done about them! Saving Earth Britannica Presents Earth’s To-Do List for the 21st Century.Britannica Beyond We’ve created a new place where questions are at the center of learning.100 Women Britannica celebrates the centennial of the Nineteenth Amendment, highlighting suffragists and history-making politicians.COVID-19 Portal While this global health crisis continues to evolve, it can be useful to look to past pandemics to better understand how to respond today.Student Portal Britannica is the ultimate student resource for key school subjects like history, government, literature, and more.From tech to household and wellness products. Britannica Explains In these videos, Britannica explains a variety of topics and answers frequently asked questions.This Time in History In these videos, find out what happened this month (or any month!) in history.#WTFact Videos In #WTFact Britannica shares some of the most bizarre facts we can find.Demystified Videos In Demystified, Britannica has all the answers to your burning questions.Britannica Classics Check out these retro videos from Encyclopedia Britannica’s archives.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
